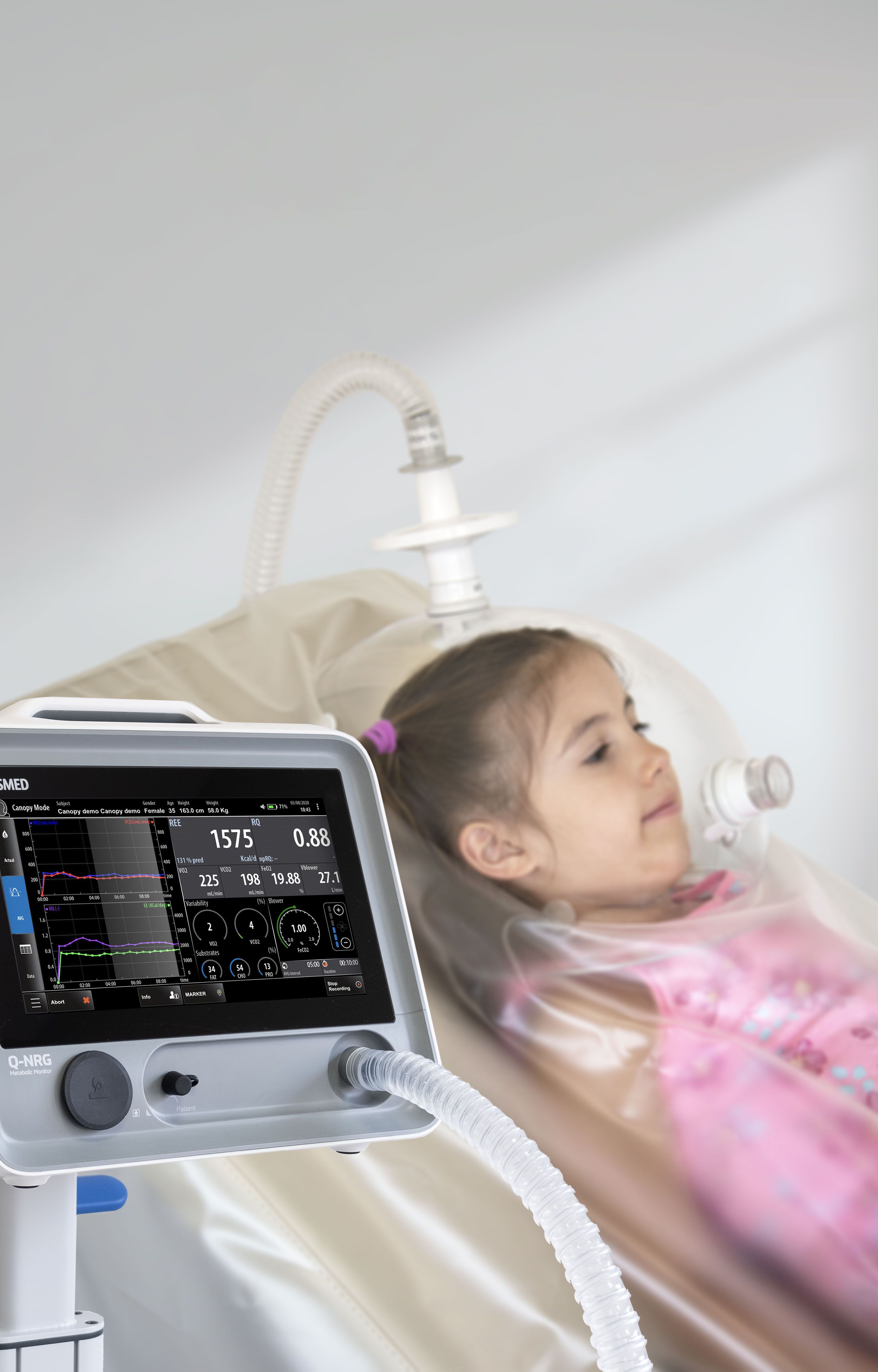
Q-NRG
The new generation of Metabolic Monitors for Indirect Calorimetry in Clinical Practice
OVERVIEW
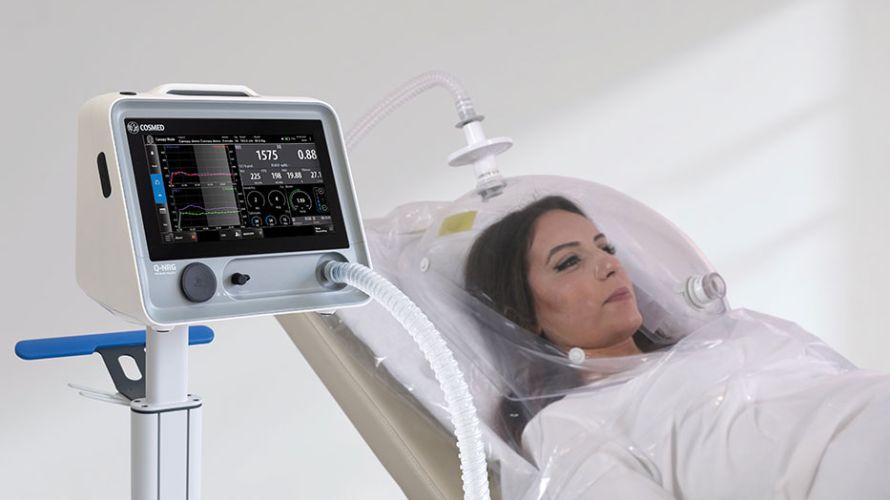
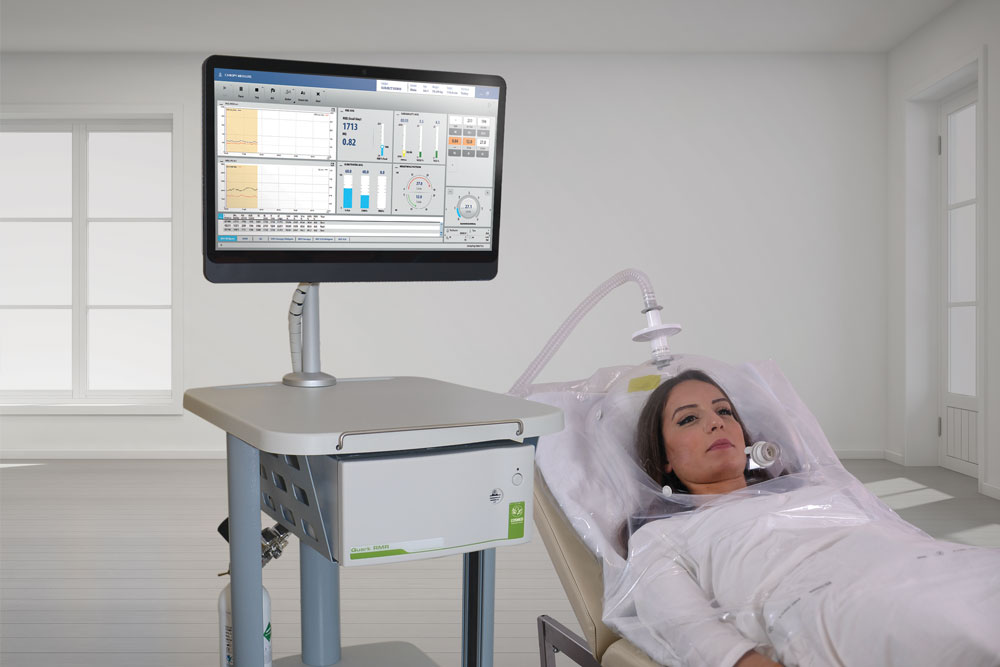
Q-NRG is the first Indirect Calorimeter specifically intended for Resting Energy Expenditure (REE) measurement on spontaneously breathing subjects.
Indirect calorimetry provides an individual and dynamic metabolic assessment based on the actual physical status of the subject rather than estimating it on anthropometric data.
Q-NRG is the result of COSMED’s collaboration with world-class institutes in the field of nutrition. Product concept and specifications have been designed together with the ICALIC Trial study group.
This collaboration made possible the development of an accurate metabolic system simple to use.
Q-NRG is a medical device.
DOWNLOAD


KEY FEATURES
Individual assessment
Q-NRG uses Indirect Calorimetry to assess metabolism. Q-NRG is the ultimate tool for research in metabolic response at resting, to develop tailored weight management programs and optimize energy balance.
Gold Standard
Q-NRG is the result of more than 30 years of experience in the design of metabolic systems. The calorimeter has been validated in-vitro and in-vivo by international multicentre studies.
Quick to operate, clean
and maintain
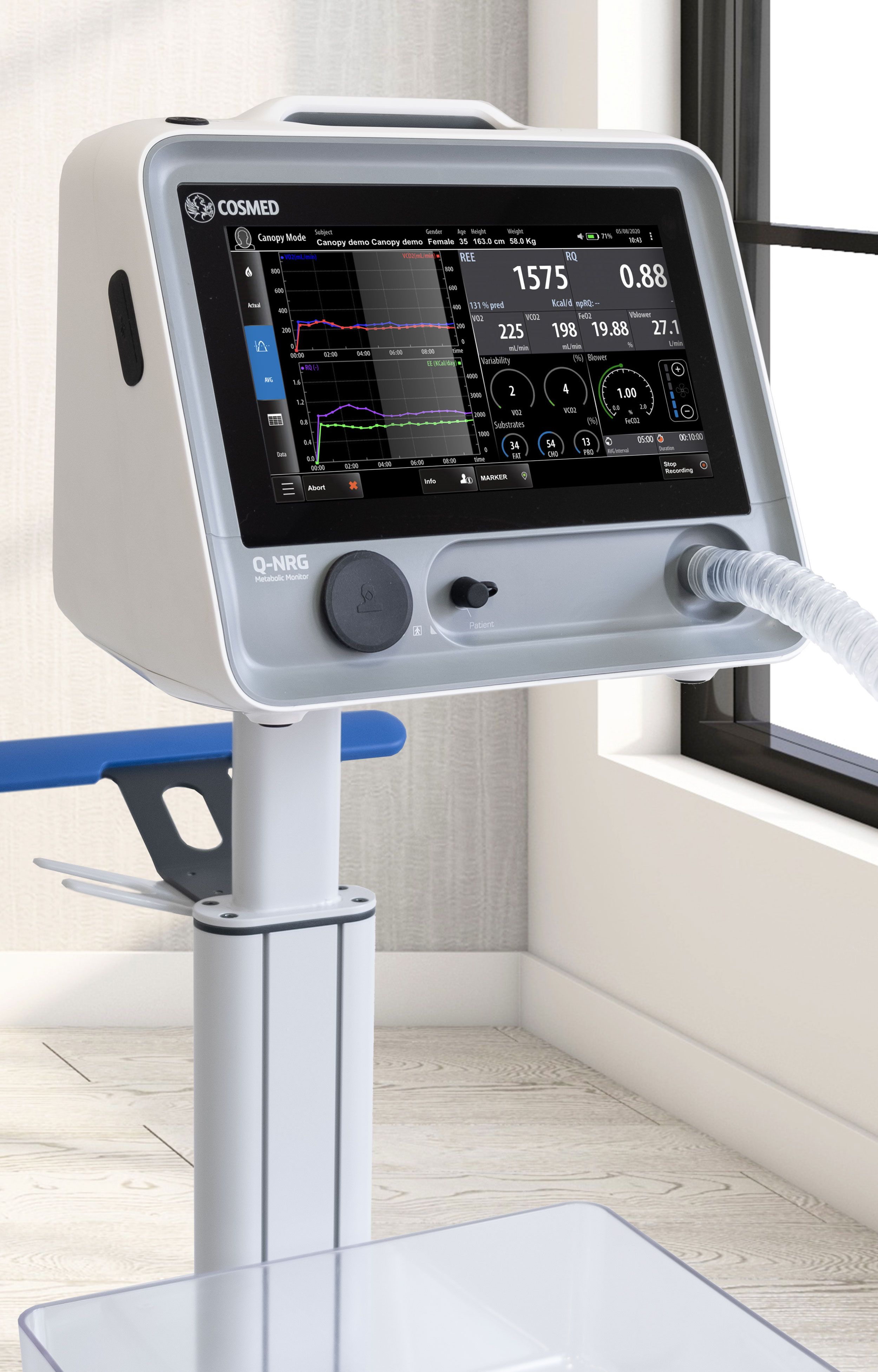
Q-NRG has been designed to reduce operations and measurement time. System does not require warm-up time nor user-assisted calibrations, all operations can be performed with a few taps on the screen.
Easy workflow
Designed for daily routine, meeting the scientific community highest standards. An intuitive workflow supports the user through all operations with main instructions prompted along the procedures.
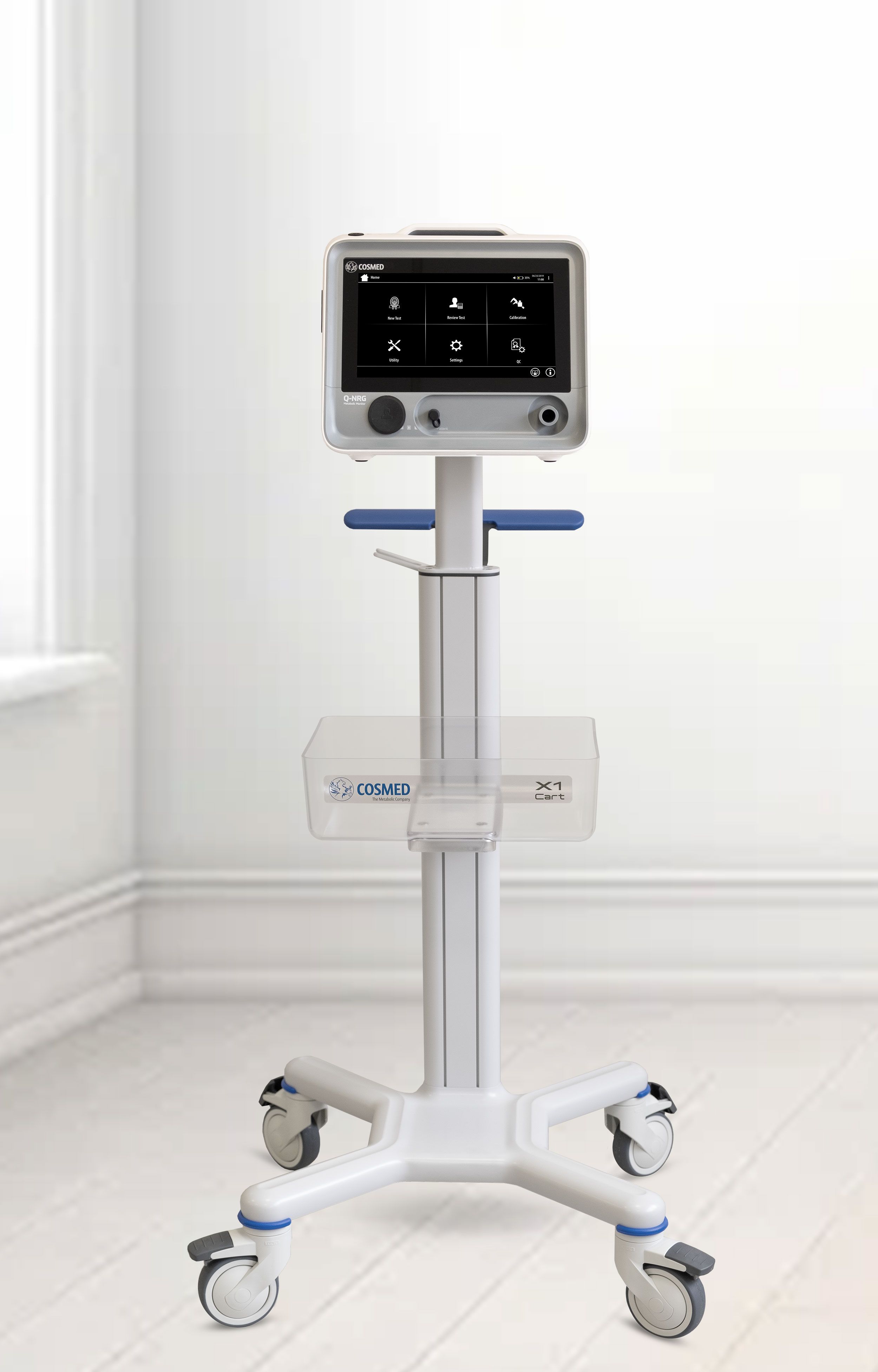
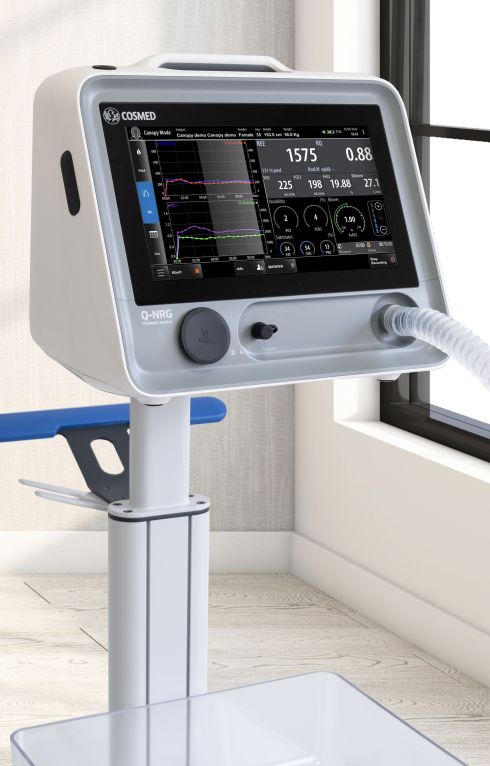
Compact
Q-NRG is a lightweight, battery operating device. The compact design guarantees extreme portability without renouncing to the comfort of a wide color LCD touchscreen.
Affordable
Q-NRG has been designed to compete with conventional metabolic systems, at a fraction of the cost.

PRODUCT INSIGHT
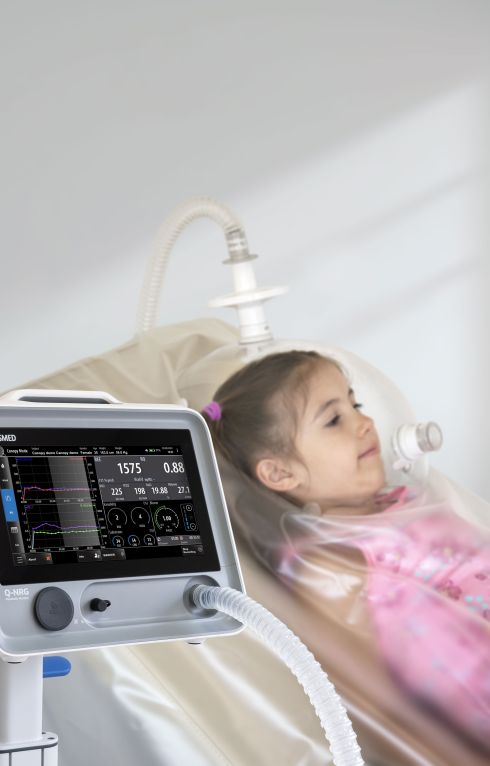
Q-NRG provides all the flexibility for a variety of clinical settings, assessing different patient's conditions and with different techniques (canopy hood and/or face masks), from adults to pediatric starting from 10 kg.


USB ports

Status LED indicators
Ergonomic handle
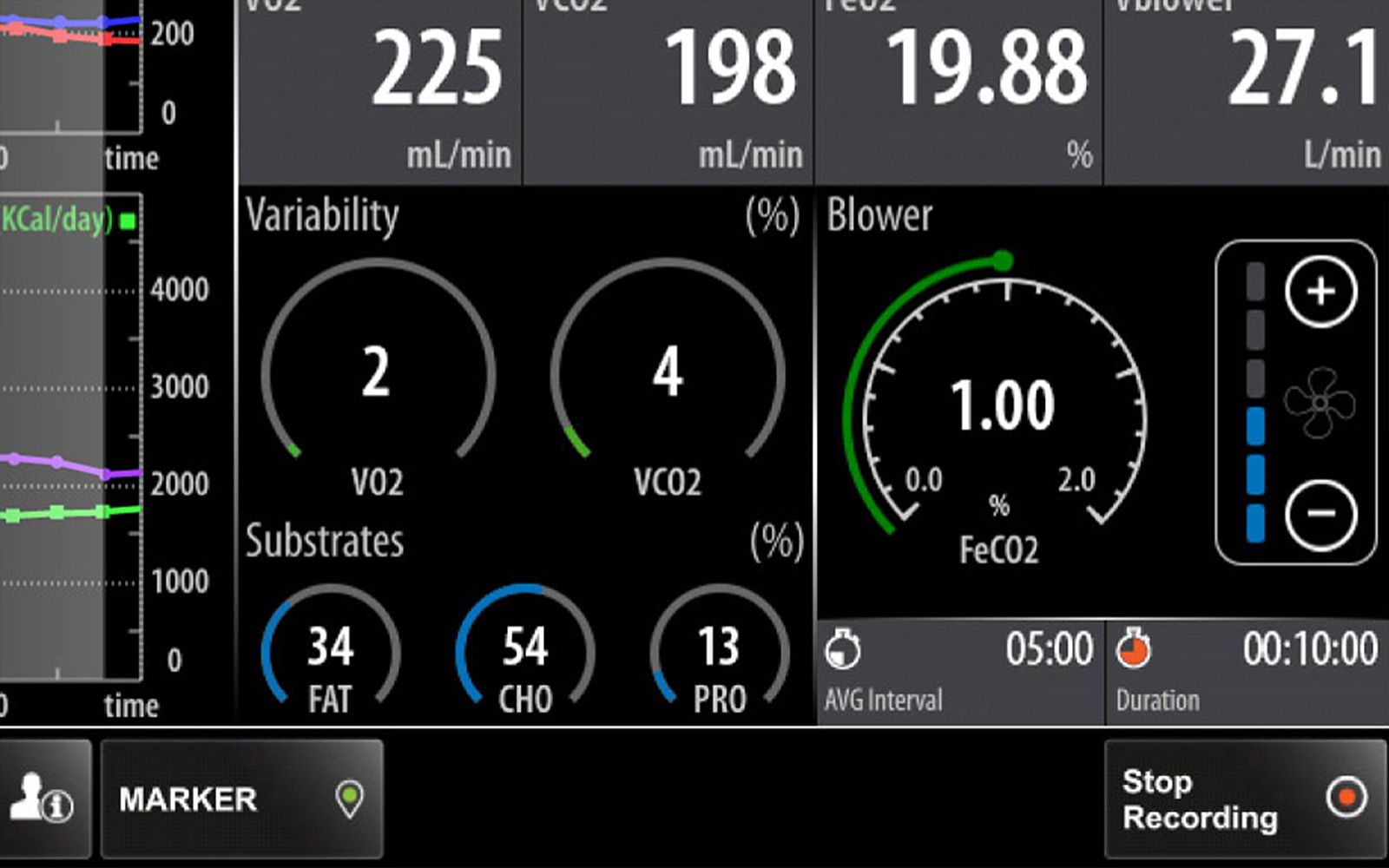
Widgets

Connections made simple


Modules
Explore the modules for the Q-NRG


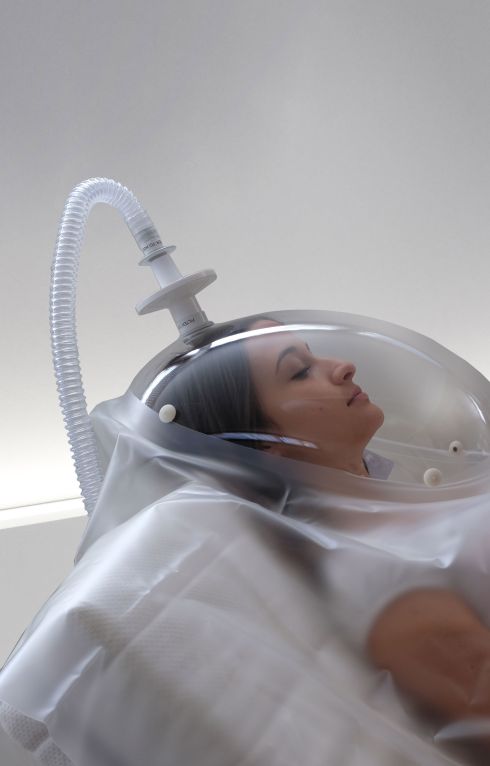
Canopy
- Measurement of oxygen consumption (VO2), carbon dioxide production (VCO2), together with other ventilatory parameters, and metabolism substrate utilization (%FAT, %PRO, %CHO).
- Spontaneously breathing subjects (>15 kg).
- Dilution technique with canopy hood (available in adult or paediatric size).
- Canopy blower pump integrated inside the unit
Mask
- Measurement of oxygen consumption (VO2), carbon dioxide production (VCO2), together with other ventilatory parameters, and metabolism substrate utilization (%FAT, %PRO, %CHO).
- Spontaneously breathing subjects (>6 yrs and >10 kg).
- Multi-use silicone face masks with headgear for comfortable leak-free measurements
- 5 mask sizes (3 adult and 2 pediatric)
Accessories
Gallery of Q-NRG accessories
Face Masks
Multi-use silicone face masks with headgear for comfortable leak-free resting and exercise gas exchange measurements
Respiratory filters
Ergonomic filters with high filtration efficiency prevent cross-contamination during all tests.


Related products
View other similar and compatible products
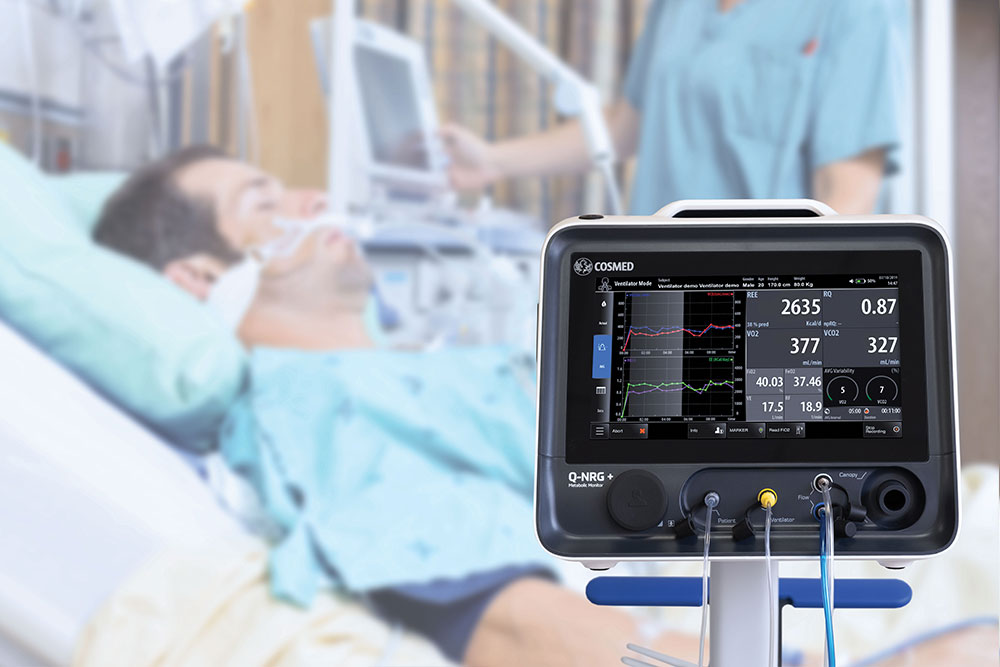
METABOLIC MONITOR
Metabolic Monitor for Indirect Calorimetry in Clinical Practice and Critical Care
METABOLIC CART
The gold standard for metabolic measurements in applied human physiology
VERIFICATION TOOL
Periodic verification of metabolic systems in clinical and research settings.


Videos
Some videos shared on COSMED's YouTube channel


Gallery





Get in touch
COSMED strives to provide the best service possible with every contact!
Fill the online forms to get the info you're looking for right now!